-Ben Chang, CEO

About Us
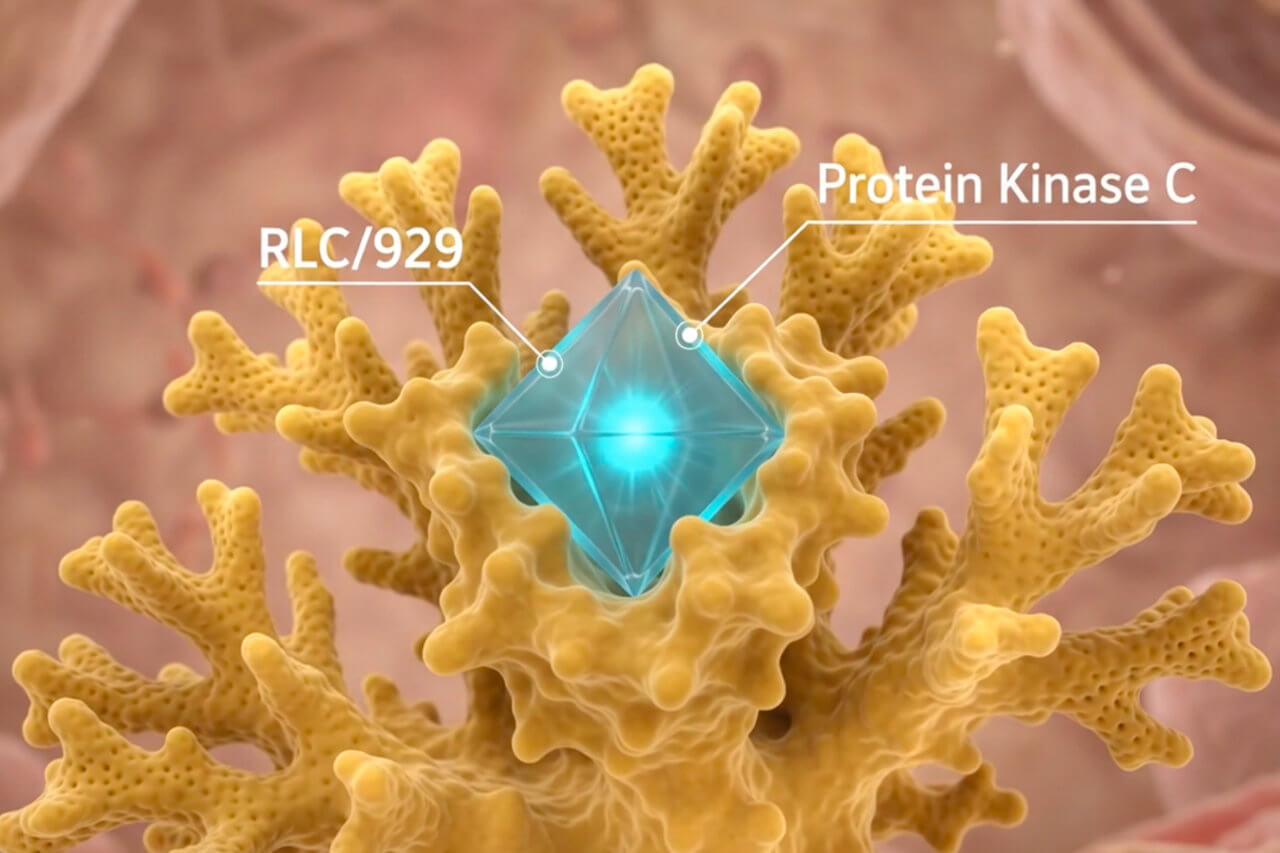
We are a biopharmaceutical company focused on the development and commercialization of RCL/929, a targeted, cell repair, immunotherapy, that promotes the reduction of inflammation for treating neurological diseases, cancer, and such inflammatory lung disorders as Acute Respiratory Distress Syndrome (ARDS).

Science and Technology
PhorMed is redefining treatment by developing cell repair therapies that restore damaged cells and reduce harmful inflammation, opening the door to new possibilities for treating cancer, brain diseases, and lung conditions. Our goal is to create safer, more effective treatments for life-threatening diseases, bringing hope to patients in need.

Patient
After multiple clinical studies RCL/929 has been found to be safe and the company is now prepared to enter into the next phase of clinical trials advancing its pipeline in collaboration with two clinical sites in Atlanta Georgia.
RECENT NEWS
PhorMed Inc is committed to good corporate citizenship. Our policy is to conduct our business affairs honestly and ethically.
03/27/2024
Pharmaron joins forces with PhorMed in ARDS studies
PhorMed continues its ARDS studies, joining forces with Pharmaron. Pharmaron, a prominent figure in drug research and development on a global scale, has acquired Absorption Systems in the USA, who PhorMed engaged for its initial ARDS study. PhorMed is excited to take advantage of Absorption Systems’ specialized expertise in scientific research and testing across various domains, including small and large molecules, cell and gene therapies, and medical devices, paired with Pharmaron’s robust capabilities. Ben Chang, CEO of PhorMed says, “We are positioned for significant growth and innovation in the healthcare sector. This partnership not only broadens our network but also grants us access to a wider array of services, underscoring our dedication to driving advancements in drug research and development.”
02/22/2024
Press Release Published announcing Equity Crowd Funding Campaign
PhorMed Inc. (PhorMed or Company) recently filed a Form C for the purpose of launching a Regulatory CF, equity crowdfunding campaign, on the platform WeFunder. For the full article click here
12/19/2023

In 2022, we completed an ARDS study and discovered that our drug has multiple pathways to at least five inflammatory markers. We were able to show that our drug, RCL/929, can reduce inflammation in the lung at two time points, 24 and 96 hours. (Our drug also reduces inflammation in the brain by more than 70%, by down regulating Aquaporin 3). We raised $700,000 from an Angel Investor to manufacture another batch of study drug, RCL/929.
In 2023, feasibility studies remain ongoing. We completed feasibility for Parkinson’s disease and started Parkinson’s preclinical study design. We completed a feasibility study for ARDS Phase 2 human trial and started the study design. We are working on a new discovery that is with our patent attorney, where he is perfecting and writing the application.
AML and HL Phase 2 protocols are final. We are now working to raise the needed funds for these clinical studies, along with the above-mentioned studies in ARDS and Parkinson’s.
